Governance & QMS
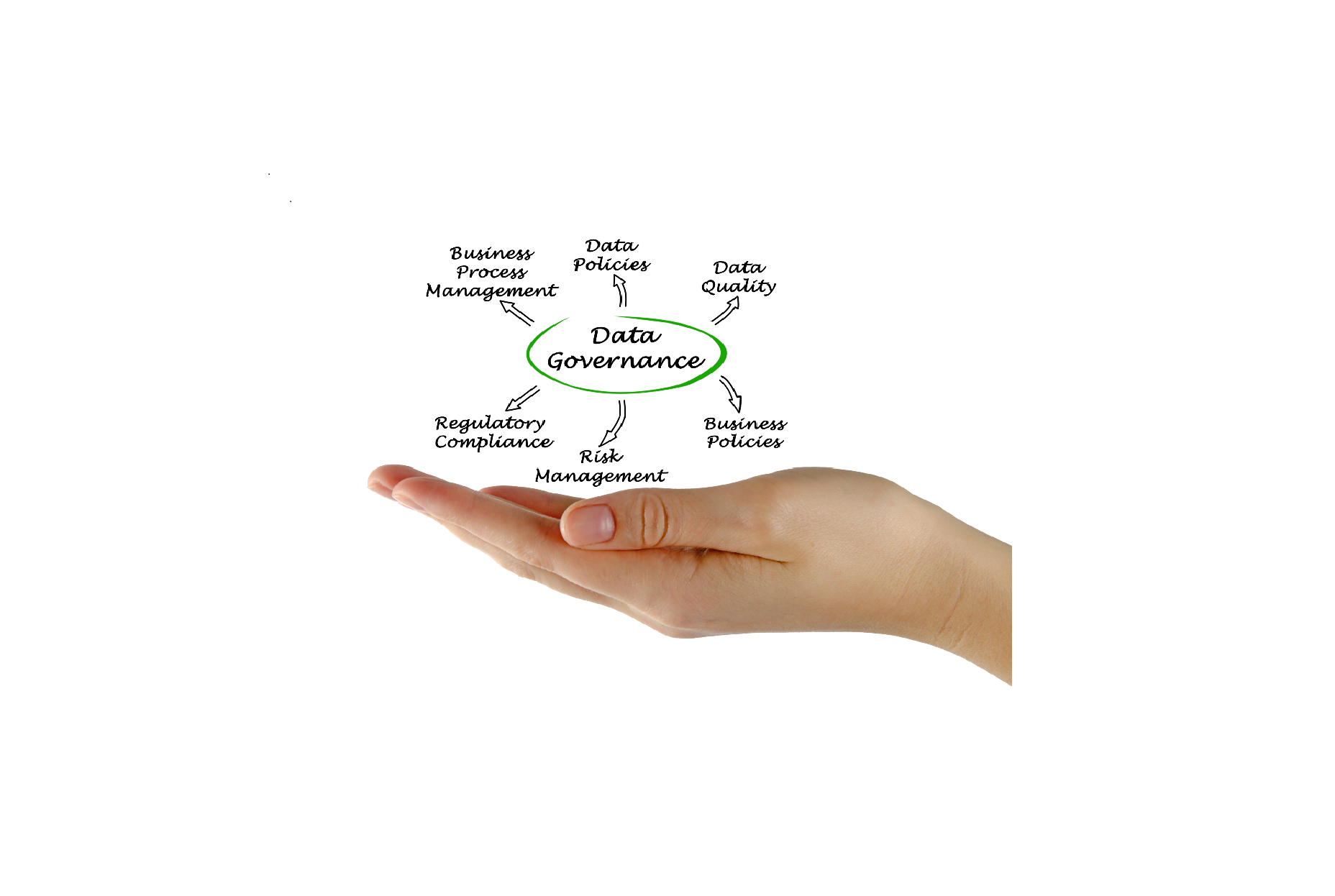
We can assess your Quality Management System (QMS) and provide Data Governance improvements and policy. It is now a requirement in the pharmaceutical industry, for any business manufacturing and/or testing pharmaceutical products, that they have a Data Governance Policy and mechanisms within the QMS to ensure efficient working and data integrity.
We can map your processes and risk assess them to find out where controls need to be inserted. We can categorise your systems and feed that back to your Data Governance Policy. We can work with you to ensure adequate Process and System ownership are implemented and maintained.
Do you need an expert pair of eyes to take a look at your QMS and ways of working to give you an objective assessment and QMS gap-analysis?. Why not get in touch and we can discuss your needs. It costs nothing for us to meet with you to scope out a way forward.
Training
We can provide training to get your people up to speed with industry legislation and expectation surrounding Validation, CSV, Data Integrity, Data Governance, and knowledge of industry practices. Do you need to know what is required of your business in light of new legislation and guidance? Learn all about MHRA and FDA legislation and guidance. Let us teach you about PIC/S, GAMP, ICH, EC and WHO guidance. We can work through some real-world examples of audit findings so you can see how you may be questioned at audit time. We can help you prepare.
Electronic Training Modules with exams are available for purchase. These training tools can be used with or without 1 or 2 day training courses held at your site or at our office.
Data Integral also offer laboratory software and Instrument training with respect to day-to-day compliant use and also the best approach for URS, DQ, Purchasing, Assessment, Configuration, Validation, Training of users, and ongoing system maintenance.
Systems
We work with you to introduce new Software and Instruments, delivering enhanced efficiency and data integrity. We ensure these new systems comply pharmaceutical industry legislation and guidance. We do this through careful URS and DQ, Risk Assessment, Functionality Assessment, Configuration, Validation, and Training.
Our people have broad experience of laboratory systems introduction ranging from simple systems to multi-Instrument network-server Chromatography Data Systems. We offer the entire introduction process from DQ and negotiation with vendors, through to configuration, CSV, procedure writing and training. You may not have in-house IT and Subject Matter Experts who can handle vendors and CSV. This is a typical and understandable situation for many Pharma/CRO/Contract Testing businesses. This is where Data Integral can come in to deliver the systems and training you need.